Cannabinoid Quantification under Ph. Eur. Monograph 3028: A Position on Correction Factors and Analytical Reliability

Our method for determining cannabinoid content in cannabis flowers is based on the European Pharmacopoeia (Ph. Eur. Monograph 3028) but has been optimized for improved performance. The contents of acidic cannabinoids (e.g. THCA, CBDA, CBNA) are reported as equivalents of their corresponding neutral forms (THC, CBD, CBN). All cannabinoid contents are expressed on a dry weight basis, as specified in Ph. Eur. 1.5.1.9. The method has been fully validated in accordance with ICH Q2(R2). Implementation of the pharmacopoeial procedure was performed in accordance with Ph. Eur. 5.26, and comparability of the modified procedure was assessed in line with Ph. Eur. 5.27.
While sample preparation follows the Ph. Eur. procedure, our chromatographic setup and quantification concept have been refined in order to improve analytical performance. In our laboratory, cannabinoids are identified by direct comparison with individual certified reference standards in each batch, rather than by relying solely on one reference substance (CBD) and relative retention times as described in the pharmacopoeial method.
Quantification is performed using five-point calibration curves for each individual reference standard (similar to the DAB monograph previously used). This eliminates the need for correction factors and avoids a source of systematic uncertainty that is inherently dependent on instrument configuration and chromatographic conditions. In our view, analyte-specific calibration provides a scientifically stronger basis for quantification than the use of fixed factors transferred across laboratories and systems.
In addition, chromatographic parameters were optimized to enable the determination of all relevant cannabinoids from a single sample solution with improved peak separation. The validated analytical range was extended to 0.1 - 40 %, whereas the pharmacopoeial procedure has been validated only up to 32 %. Taken together, these modifications make the method more robust, more efficient, and analytically more reliable, while improving identification, quantification, and operational practicality.
Position on correction factors
The pharmacopoeial procedure uses a single calibration reference (CBD) together with correction factors to calculate the contents of THC(A), CBDA, and CBN(A). These correction factors are derived from relative response factors (RRFs), i.e. the detector response of individual cannabinoids relative to CBD.
Because these response factors depend on the analytical system and chromatographic conditions applied, fixed correction factors cannot be assumed to be universally transferable. For that reason, laboratories using this quantification concept should establish and verify laboratory-specific correction factors under their own operating conditions. In our view, this constitutes a relevant analytical procedure performance characteristic (APPC) and should be assessed accordingly in line with Ph. Eur. 5.26.
The determination of laboratory-specific correction factors requires measurement of peak areas from standard solutions across multiple concentration levels, followed by linear regression and calculation from the resulting slopes:
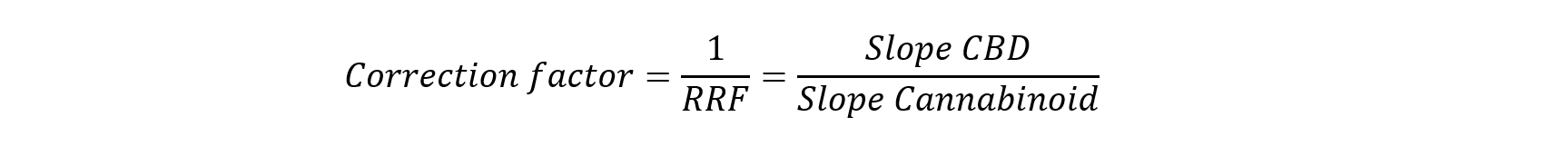
Using this approach, the relative response factors for the relevant cannabinoids were determined in our laboratory. The resulting empirical correction factors deviated by up to 12 % from those stated in the Ph. Eur. monograph (see Table 1).
Table 1: Comparison of Correction Factors
| Cannabinoid | Correction factors QSI | Correction factors Ph.Eur. | Percentage deviation |
| CBD | 1 | 1 | |
| CBDA | 0.537 | 0.596 | -9.9% |
| THC | 1.112 | 1.097 | 1.4% |
| THCA | 0.607 | 0.691 | -12.2% |
These deviations, particularly for acidic cannabinoids such as THCA, CBDA, and CBNA, have a direct impact on the reported total cannabinoid content. In a comparative assessment using identical chromatographic data from a 28/1 cannabis variety, quantification based on the unverified Ph. Eur. correction factors produced higher results than quantification based on individual certified reference standards (see Figure 1). This demonstrates that THCA, and consequently total THC, can be overestimated when fixed correction factors are applied without laboratory-specific verification. In practical terms, this creates a real risk that products which are analytically compliant may appear to fall outside the permitted specification range.
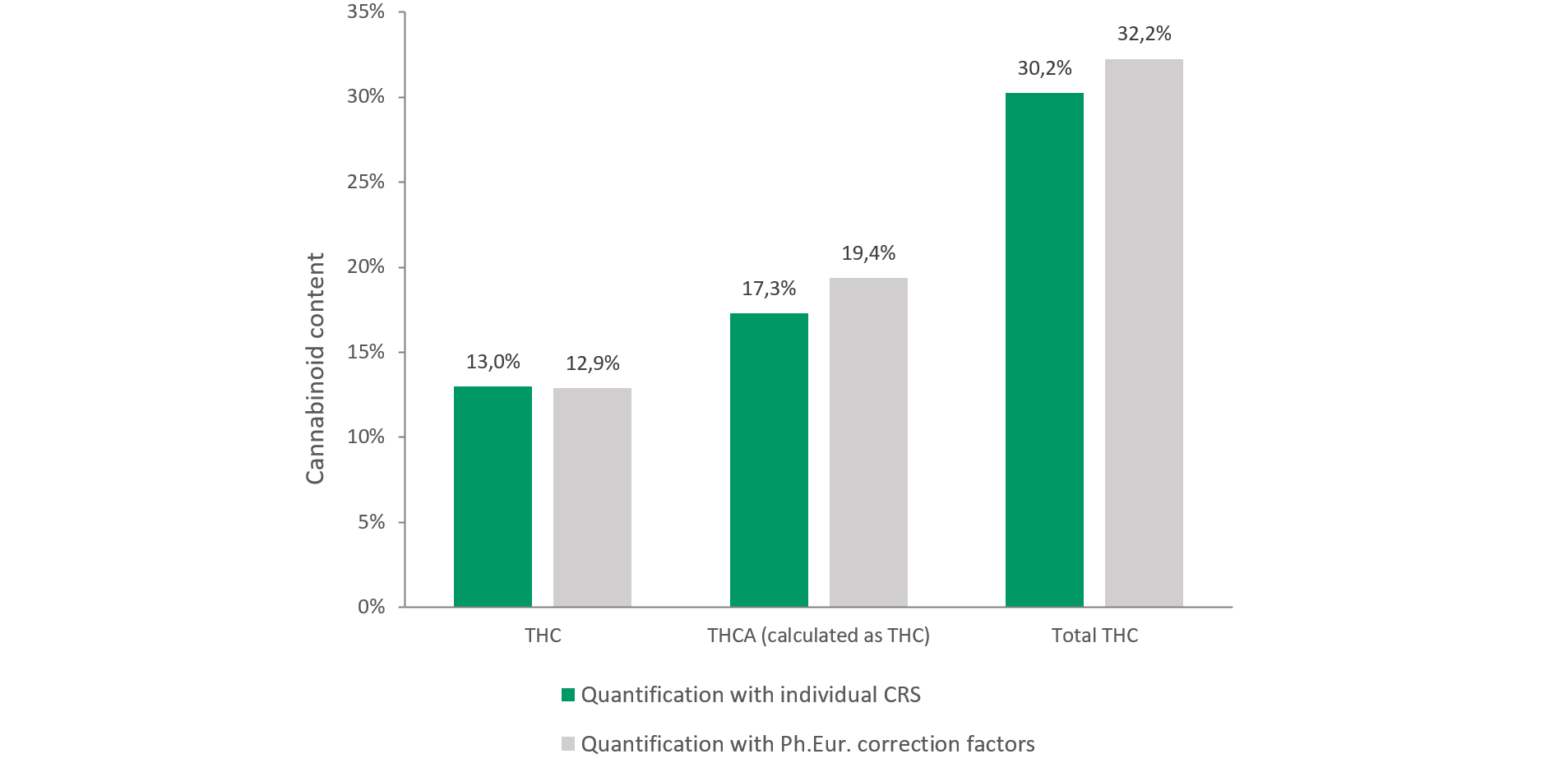
Figure 1: Exemplary comparison of THC and THCA quantification in a representative cannabis flower
This is not merely a theoretical concern. Very similar discrepancies have been observed by several independent laboratories, underscoring that fixed correction factors are not universally applicable, particularly not for the assay of active substances subject to a permitted deviation of no more than ±10 %.
According to general chapter 2.2.46 of the Ph.Eur., correction factors are primarily intended for impurity or related substances testing and should only be applied when response factors differ markedly. Applying such factors to the quantification of active cannabinoids is scientifically inappropriate, as it introduces avoidable uncertainty into the assay of main active substances total THC and total CBD.
For this reason, fixed correction factors should not be adopted without prior verification under laboratory-specific conditions. Laboratories should either establish and validate their own correction factors for their own system or, preferably, quantify each cannabinoid using individual certified reference standards. This approach provides a more direct, more transparent, and more scientifically robust basis for quantification. This is also consistent with Good Analytical Practice and the principles of quantitative method validation described in ICH Q2 (R2).
We recognize that the practical implementation of Ph. Eur. 3028 continues to raise questions in routine laboratory practice and in the comparison of results across laboratories. By sharing this position, we aim to contribute constructively to this discussion and to support transparent, scientifically robust approaches to cannabinoid quantification. We are always available for questions, technical exchange, and further discussion with customers, laboratories, and regulatory stakeholders.
We welcome further questions and discussion. Please feel free to contact us:
P: +49 421 59 66 07-40